The Path to Commercializing Rare Bioactives

The demand for cleaner, more sustainable, and cost-effective bioactives across all industries has never been higher. Consumers want the option to “do better” – for themselves, others, and the planet.
Companies are finding themselves in a new age of innovation where development of new, rare bioactives that deliver on promises is key to connecting with consumers. But finding and bringing these ingredients down the long and winding path to market is no small feat. It requires a strategic selection of markets, fast scale-up, and a keen eye on regulation.
“Nature is rich with bioactives that people have relied on for centuries,” says Cynthia Bryant, Chief Business Officer at Demetrix. “Oftentimes though, they are found in very low quantities in their botanical sources, making it difficult to harness their potential in consumer products and pharmaceuticals.”
And Ms. Bryant would know this well. Over the last 15 years, she has been completely focused on commercializing bioactives produced through biotechnology, holding senior leadership roles in companies producing high-quality ingredients through fermentation. Her specialty is planning go-to-market strategies and identifying the right consumer products or pharmaceuticals that can benefit from increased sustainability, supply, or purity.
To better understand the hurdles of developing innovative and promising ingredients, such as the rare bioactives being developed and produced by Demetrix, we sat down to chat about the process of commercialization.
Q: How does the commercialization process Demetrix is navigating differ from those of your typical biotech company?
When I think about the ingredients biotech has been focused on over the decades, there is one common thread – they are using their technology to produce ingredients already available in various markets. For example, nutritional ingredients, flavors like vanilla, and fragrances.
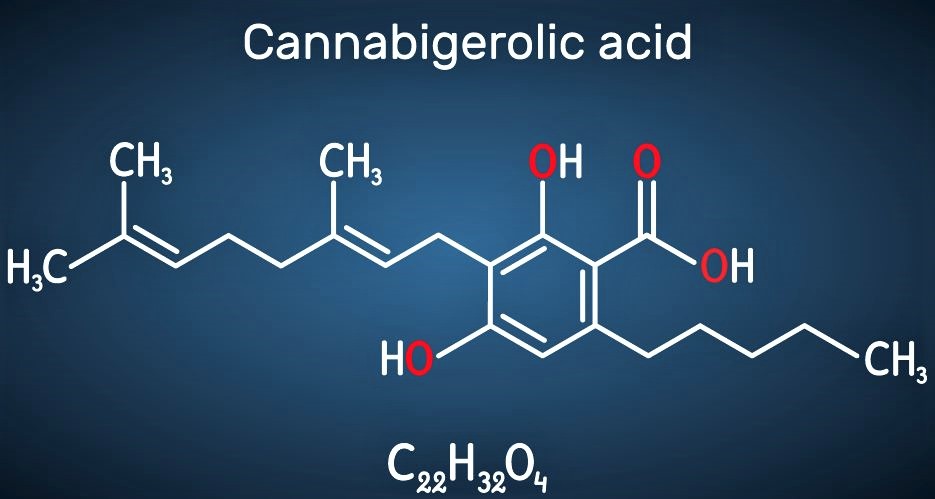
What’s different at Demetrix is that we’re focusing on a class of compounds that are not commonly used today. And it’s because of the lack of access that’s been going on for decades. But, they’re super interesting and there’s evidence pointing to the fact that they have beneficial qualities for people and animals. They just haven’t been researched, and because of this, they haven’t been fully utilized for health and wellness. This class of compounds are cannabinoids.
So, that’s a key difference for us. First, we’ve only scratched the surface of what cannabinoids can do and we need to clearly understand the unique properties of these ingredients and how that can translate into benefits for various consumer products, whether it’s a topical skin cream or a daily supplement or something you put in a sports drink.
Second, we need to educate prospective industries on why specific cannabinoids are compelling and should be included in the formulations of their products.
Q: How is what Demetrix is doing similar to the typical commercialization process a biotechnology company might take?
We’re taking a very traditional path that all biotech companies take, which is the progression of developing the technology and scaling it up to be able to produce the ingredients at an affordable cost to use in consumer products.
For scale-up, a lot of it is focusing on bench scale, also known as a proof-of-concept. A lot of people can make things work at bench scale. The real art is scaling from a one to 10-liter tank on a benchtop to a ~100,000 liter, multi-story tank. The right expertise is needed to successfully navigate this process.
This is where the experience of our team comes into play. Arguably, the Demetrix team collectively has successfully commercialized more ingredients using this technology than any other company in this space. It’s this experience that makes the difference.
Q: How do you balance your scale-up considerations with all of the other commercial considerations?
Well, if we’re going to sell our product as a supplement or a food ingredient, we need to run important tests to ensure it is safe. But those tests have to be run on a product that uses our end process, so we are very careful about when we start that testing. You don’t want to start it too early, then change your process and void the tests and have to do them over again.
The balance is between R&D, scale-up, regulatory, and the business. We have to get all four of these lanes and their respective milestones synchronized, so at the end, we have an ingredient that’s at the required specification, at the quantities needed, for the supply agreements we’ve signed, in the markets that we know are safe to sell our product.
Q: How do you define which market or markets you want to be in, especially with cannabinoids, an ingredient that could be fundamental for so many industries?
This is probably the most interesting aspect of what we do each day. And that’s because our research indicates cannabinoids can be applied to many different aspects of improving health and wellness.
They show potential for topical applications to improve skin health, including microbiome support, antioxidant, or anti-inflammatory properties. We also see indications to help with anxiety, stress, immune support, and sleep aids. Then there are the pharmaceutical applications which are starting to emerge.
We use the insights from our research to identify what markets would be best served by each cannabinoid. We then work to clear all regulatory requirements to ensure they are safe and legal to use in each target market.
Q: What step is Demetrix currently at in the commercialization process?
Right now we’re scaling up with our commercial partner. We anticipate that we will be at commercial production fairly soon and are working with our various partners to get ready for that.
Q: How do you define personal and professional success throughout this commercialization process?
For me personally, what’s going to be very satisfying is to see products that are available to people that hold true to their promise to help with whatever issue it is that they might have. Whether it’s a reduction in skin redness or helping with sleep issues or digestive support.
I believe this going to be the highlight of my career, to know that I had an active part, and Demetrix as a whole had an active part, in ensuring these bioactives get to market in a safe manner, a legal manner, and also in a way that allows them to deliver efficacious results for people.
Q: How does Demetrix’s progress line up with those of your competitors?
For me, it’s more about engaging with the prospective customers in the targeted markets that we’re after and educating them about the true benefits of the cannabinoids we are bringing to market. I tend to hyper-focus on building markets and providing access to people that need help.
I’m excited to see a lot of us succeed in this industry because if all of us are succeeding, it proves the point that these are ingredients that help people.
Q: If there is one thing about cannabinoids you would want people to walk away from this interview knowing, what would it be?
I feel that we’re on the verge of something super interesting from a health and wellness perspective. Cannabinoids are a class of compounds that have been completely skipped over by modern science and we know at least from the data that we’ve seen that they have an interesting safety profile. We know a few of them have beneficial effects on some pretty important indications.
I’m very eager to see this industry and the use of these ingredients grow. I hope to see people thrive in ways that they weren’t able to before and that other people share my enthusiasm because I do believe this class of compounds can be a significant benefit to a lot of people.